 [
[

Prof. Liu Cong from the Shanghai Institute of Organic Chemistry of the Chinese Academy of Sciences, along with collaborators, has revealed the high-resolution structures of amyloid fibrils directly extracted from biopsy samples of living patients with systemic light chain (AL) amyloidosis, providing new molecular insights into how patient-specific protein sequences and tissue environments shape amyloid architecture.
The findings are published in the journal Proceedings of the National Academy of Sciences.
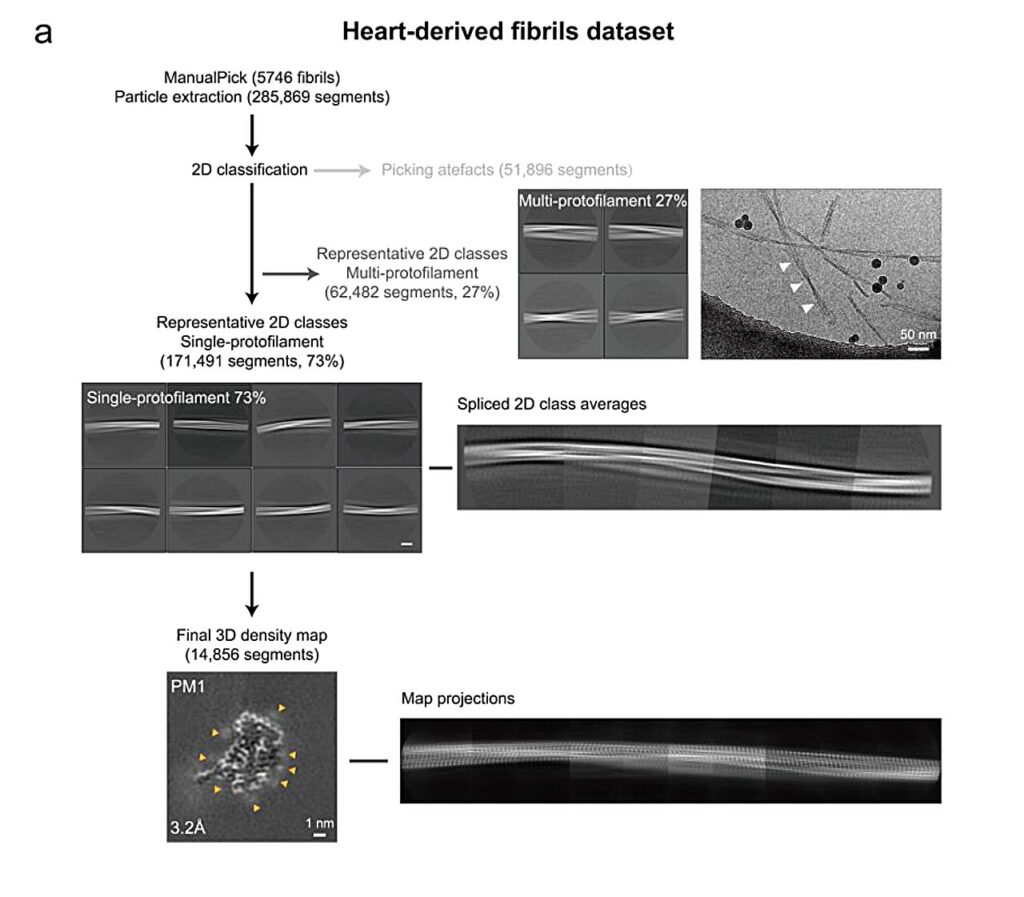
The researchers analyzed amyloid fibrils isolated from abdominal fat and cardiac biopsies obtained from three AL amyloidosis patients. Using cryo-electron microscopy (cryo-EM), they resolved five distinct atomic structures of immunoglobulin light chain-derived fibrils. The fibrils were derived from clinically accessible biopsies of living patients instead of postmortem or explanted tissues, providing a closer view of disease-relevant amyloid structures.
The researchers found pronounced patient-specific structural diversity of amyloid fibrils. Fibrils from different patients adopted distinct atomic conformations, even when derived from the same immunoglobulin light chain germline family. This finding highlights the dominant role of individual protein sequences and patient-specific variations in defining fibril architecture.
In contrast, the researchers found that amyloid fibrils extracted from different tissues within the same patient, notably abdominal fat and heart, exhibited highly conserved global folds. Although there were subtle local differences, including side-chain rearrangements and additional densities near the fibril surface, the overall fibril structures remained highly similar. These localized variations are likely influenced by tissue-specific microenvironments and associated molecular cofactors.
Abdominal fat biopsy is widely used as a minimally invasive diagnostic tool for AL amyloidosis, yet it has remained unclear whether fibrils obtained from fat accurately represent those deposited in vital organs such as the heart. The structural similarity revealed in this study provides direct molecular evidence supporting the diagnostic relevance of fat-derived amyloid fibrils.
Moreover, the researchers identified conserved aggregation-prone regions, disulfide bonds, and posttranslational modifications within light chain fibrils. They frequently observed additional non-proteinaceous or protein-associated densities adjacent to aromatic residues, suggesting that extracellular matrix components or other tissue-derived cofactors may participate in fibril stabilization and polymorphism.
By integrating cryo-EM, mass spectrometry, and biochemical analyses, this study provides a biopsy-resolved structural framework for understanding AL amyloidosis. It demonstrates how intrinsic protein sequences and extrinsic tissue environments jointly determine amyloid structure, paving the way for improved diagnostic strategies and the development of amyloid-targeting therapeutics.
Publication details
Yuxuan Yao et al, Biopsy-resolved cryo-EM structures of amyloid fibrils provide molecular insights into AL amyloidosis, Proceedings of the National Academy of Sciences (2026). DOI: 10.1073/pnas.2515454123
Journal information:
Proceedings of the National Academy of Sciences
Key medical concepts
Clinical categories
Citation:
Biopsy-derived cryo-EM structures reveal patient-specific amyloid fibrils (2026, January 13)
retrieved 13 January 2026
from https://medicalxpress.com/news/2026-01-biopsy-derived-cryo-em-reveal.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.