 [
[
Seventy-five percent of patients with blastic plasmacytoid dendritic cell neoplasm (BPDCN) who were treated with the antibody-drug conjugate (ADC) pivekimab sunirine (PVEK) had a complete response, according to new data from the Phase I/II multicenter international CADENZA trial led by researchers at The University of Texas MD Anderson Cancer Center.
The findings were published in the Journal of Clinical Oncology. The study was led by principal investigator and corresponding author Naveen Pemmaraju, M.D., professor of Leukemia, and senior author Naval Daver, M.D., professor of Leukemia.
“These strong, durable response results offer hope to BPDCN patients with limited treatment options,” Pemmaraju said. “An effective and safe frontline treatment for patients would be practice-changing, and these positive results suggest that PVEK should be considered a potential standard treatment for BPDCN patients.”
Early data from the trial were presented at the 67th American Society of Hematology (ASH) Annual Meeting and Exposition. In addition, data were shared at the 2025 American Society of Clinical Oncology Annual Meeting.
Understanding a rare aggressive cancer
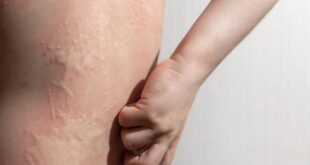
BPDCN is a rare, aggressive blood cancer that affects multiple organs, including a patient’s skin, bone marrow, and lymph nodes, which makes diagnosis and treatment complex. The disease is difficult to treat because of its hybrid nature, having elements of both lymphoid and myeloid malignancies, according to Pemmaraju. In addition, the disease can frequently mimic other related blood cancers or present alongside them.
The current standard of care for patients with BPDCN includes treatment with tagraxofusp-erzs, which targets CD123 found at high levels on certain cancer cells, including BPDCN. In 2018, researchers from MD Anderson led the clinical development to advance tagraxofusp-erzs toward Food and Drug Administration approval.
PVEK is a next-generation CD123-targeted treatment. CD123 is overexpressed in the disease, leading to other therapeutic targets not working. PVEK delivers the therapeutic drug directly to cancer cells by targeting CD123 on the surface of BPDCN cells, resulting in rapid cancer cell death with limited targeting of healthy tissue.
Trial design, responses and survival
A total of 84 patients were treated as part of the trial. Of these, 33 patients received PVEK as a frontline treatment and 51 had relapsed or refractory disease. Eleven patients in the frontline treatment group had prior or concurrent cancer diagnoses in addition to BPDCN, making their treatment more complex and challenging.
For the frontline-treated patients, the overall response rate was 85%, and the median overall survival was 16.6 months. Eight of these patients proceeded to a stem-cell transplant, which is the only established curative option for BPDCN and is generally associated with improved survival in BPDCN. Patients with relapsed or refractory disease had an overall response rate of 35% and a median overall survival of 5.8 months.
Safety profile and future directions
Side effects were manageable overall, including peripheral edema and infusion-related reactions. The study’s overall efficacy suggests that PVEK could offer a valuable new option for patients with difficult-to-treat BPDCN.
Researchers at MD Anderson also are investigating the use of PVEK in combination approaches to treat frontline acute myeloid leukemia (AML).
“We are encouraged by what we are seeing in the treatment of BPDCN, and early results also indicate encouraging efficacy in frontline AML, including in combination approaches, as presented at the 2025 ASH Annual Meeting,” Daver said.
Publication details
Naveen Pemmaraju et al, Pivekimab Sunirine in Blastic Plasmacytoid Dendritic Cell Neoplasm, Journal of Clinical Oncology (2026). DOI: 10.1200/jco-25-02083
Journal information:
Journal of Clinical Oncology
Key medical concepts
Clinical categories
Citation:
Antibody-drug conjugate achieves high response rates as frontline treatment in aggressive, rare blood cancer (2026, February 12)
retrieved 12 February 2026
from https://medicalxpress.com/news/2026-02-antibody-drug-conjugate-high-response.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.